UNLOCKING THE POTENTIAL OF THE
IMMUNE SYSTEM WITH A NEW
CLASS OF THERAPEUTICS
Immunophotonics uses a proprietary glycan polymer with
platform potential to stimulate antigen capture and uptake along
with other immunological processes that are intended to trigger an
immune response, thereby enabling the rejection of cancer and other diseases.
Read More
Unlocking the potential
of the immune system
with a new class of
therapeutics
Proprietary glycan polymer with platform potential
stimulates antigen capture, uptake, and
immunological responses.
Intended to stimulate an immune response, thereby
enabling the rejection of cancer and other
diseases.
Practical Applications to Elevate Modern Medicine
Transforming
Interventional
Oncology
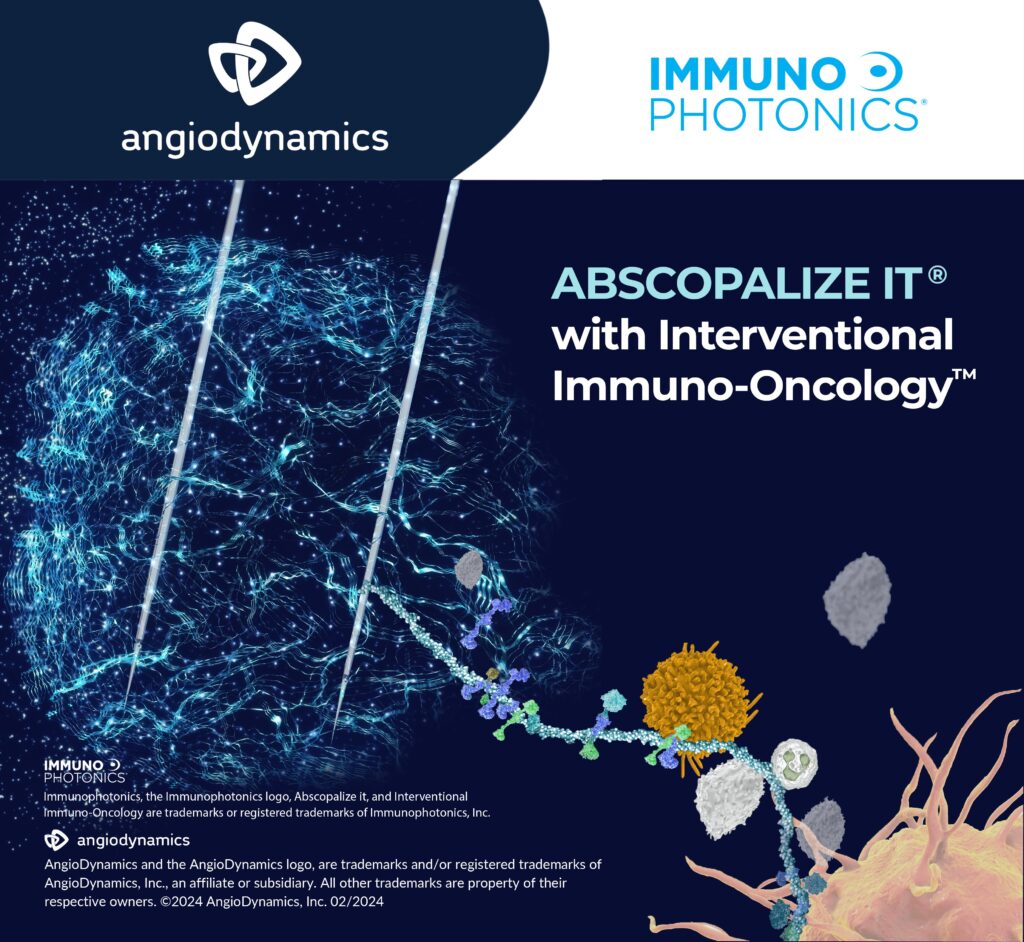
Injecting IP-001 following routine tumor ablations may transform this conventional surgical intervention into a systemically active immunotherapy.
Elevating
Immunotherapy
IP-001 upregulates T-cell activation, downregulates T-cell exhaustion, and drives stronger Th-1 and cytotoxic T-cell response, which could enhance other immunotherapies, such as checkpoint inhibitors.
Boosting
Vaccines
IP-001 is a powerful immunoadjuvant that enhances antigen-specific immunity against infectious diseases, including SARS-CoV-2, potentially increasing the efficacy of vaccines.
Immunophotonics is dedicated to developing its proprietary technology for the benefit of global human health. We believe our multimodal approach can elevate modern medicine, allowing everyday-routine treatments to provide enhanced outcomes.