Immunophotonics Switzerland (IPS Biopharma) will be presenting at the Swiss Biotech Day in the Emerging Company session hosted by BB Pureos Bioventures. The Swiss Biotech Day is one of the premier annual biotechnology conferences in Europe, featuring panel discussions and presentations from key opinion leaders in the biotech industry. Immunophotonics’ presentation will take place on...
Category: Latest News
Immunophotonics Publishes Follow-up Feature in Nature Biopharma Dealmakers
Immunophotonics is excited to be featured in the September 2021 oncology issue of Nature Biopharma Dealmakers. Read what Dr. Markus Joerger and Dr. David Anderson, distinguished clinical and scientific experts, have to say about how Immunophotonics is pioneering the growing field of Interventional Immuno-Oncology (IIO) with its lead drug candidate, IP-001. To read the article,...
Immunophotonics Featured in Nature Biopharma Dealmakers
Immunophotonics has been featured in the March oncology issue of Nature Biopharma Dealmakers (https://www.nature.com/articles/d43747-021-00020-2 ). Nature selects only a handful of companies for such articles. The article highlights our uniqueness in pioneering Interventional Immuno-Oncology and conveys some of our recent findings.
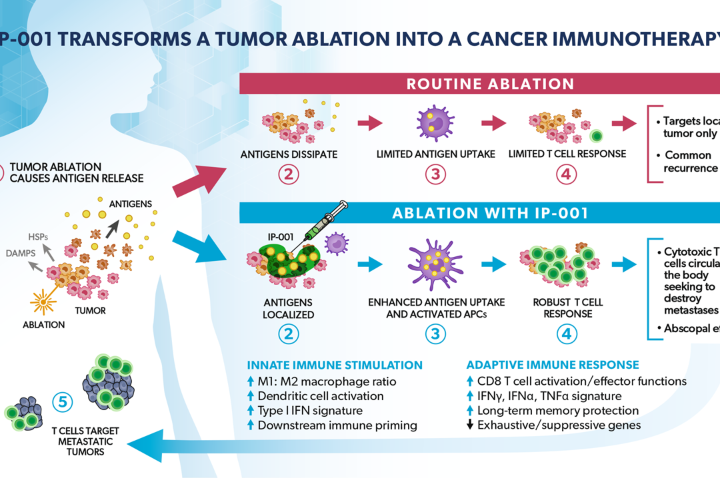
Study Shows IP-001 to Amplify Tumoricidal and Immune Effect of Cancer Ablation Therapies
A paper recently published in Cells has described how Immunophotonics’ immune-stimulating drug IP-001 was shown to be effective against metastatic tumors when combined with different forms tumor ablation. In this review, the authors briefly discuss the current applications of local ablation for cancer treatment and the effects of IP-001 in combination with other ablation therapies,...
Immunophotonics Announces First Patient Treated in Swiss Clinical Trial
ST. LOUIS and BERN, Switzerland, Nov. 10, 2020 (GLOBE NEWSWIRE) — Immunophotonics, Inc. announces the first patient treated in the phase 1b/2a clinical trial examining IP-001 in thermally ablated solid tumors. The trial, conducted in collaboration with the Swiss Group for Clinical Cancer Research (SAKK), will assess the safety, tolerability and anti-tumor activity of IP-001...
Potential COVID-19 Applications of Immunophotonics’ Leading Drug Candidate, IP-001, Being Researched at the University of Oklahoma
OU Daily at the University of Oklahoma recently published a story on research being conducted by Dr. Wei R. Chen on IP-001’s potential application as an immune-system booster that could be used in conjunction with a COVID-19 vaccine. The story is available here.
Immunophotonics Announces Appointment of Jonathan Knowles to Board of Directors
October 05, 2020 04:13 ET | Source: Immunophotonics, Inc. BERN, Switzerland, Oct. 05, 2020 (GLOBE NEWSWIRE) — Immunophotonics, Inc., a biotechnology company focused on the discovery and development of proprietary immune-activating carbohydrate polymers for the treatment of solid and metastatic cancers, has bolstered its expertise through the appointment of Jonathan Knowles, Ph.D. to its Board...
Immunophotonics Receives Swissmedic Approval to Commence Phase 1B/2A Clinical Trial in Switzerland
Immunophotonics has received Swissmedic approval to commence a new clinical trial entitled “Intratumoral Injection of IP-001 Following Thermal Ablation in Patients With Advanced Solid Tumors.” This is a multicenter phase 1B/2A trial with expansion cohorts in melanoma and soft tissue sarcoma patients. The therapeutic approach taken by this trial (SAKK 66/17) is different from those...
Immunophotonics and Clinical Laserthermia Systems Announce Immuno-Oncology Research Collaboration and Clinical Trials for Cancer Patients with Solid Tumors
Collaboration will Combine Immunophotonics’ Proprietary Drug, IP-001, and CLS’s TRANBERG Laser Thermal Therapy System and imILT Method ST. LOUIS and IRVINE, Calif., June 14, 2019 (GLOBE NEWSWIRE) — Immunophotonics, Inc. and Clinical Laserthermia Systems, AB (STO:CLS B) (CLS) today announced they have entered into a research collaboration agreement to support a Phase 1b/2a clinical trial...